
Potassium-based compoundsĪll potassium compounds are ionic in nature: It is the most common form in which potassium occurs in nature. Potassium ions, with the formula K +, are positive monatomic ions, also called cations, carrying a positive excess charge. Potassium is a very light, silvery white alkali metal, and it is very reactive with water.įor plants, the potassium ion plays an important role in maintaining osmotic pressure and in certain metabolic processes such as plant growth. It comes in a metallic form which can be obtained by electrolysis but which does not exist in nature. It is a greyish metal, very soft, ductile and above all very reactive since it is one of the most reducing metals. Physical and chemical properties of Potassium Potassium as a single body Polyhalite- K 2 Ca 2 Mg (SO 4 ) 4 2H 2O.There are many minerals composed of potassium such as: It is moreover this process which was discovered and used by Sir Davy. However, potassium is not a native element since it is obtained, at least mainly, via the electrolysis of potassium hydroxide in a molten dry process. Indeed, its Clarke is high, it represents 2.58% of the total weight of the earth's crust, making it one of the 7 most abundant elements on Earth. Potassium is a very abundant element in the earth's crust. Potassium Ion K - electron configuration: 1s 22s 22p 63s 23p 64s 2ĭiagram of Potassium electron configuration.Electronic configuration of potassium Ion K + : 1s 22s 22p 63s 23p 64s 0.Potassium has total 19 electrons, we fill the electron shells as below: The potassium atom (K) and Cl -2, Sc +2, Ti +3, V +4, Mn +6 have the same electronic configuration. Also you can write potassium electron configuration in abbreviated form i.e. Potassium, with atomic weight 39.098 and atomic number 19, has the fundamental electronic configuration of 1s 22s 22p 63s 23p 64s 1. Electronic configuration of Potassium (K) Potassium was the first metal isolated by electrolysis, the decomposition of a chemical body under the effect of an electric current. Some compounds, such as potash, have been known and used since a very old time but potassium was not identified as an element until 1807, when the British chemist Humphry Davy carried out the electrolysis of molten potassium hydroxide and obtained a still unknown metal. So now question is- Who discovered Potassium? The name potassium was proposed in 1807 by the discoverer of this element. Regarding the symbol "K" it derives from the German name of this element "kalium". It should be noted that at that time the term potash designates calcium carbonate while today it corresponds to a solution of potassium hydroxide. Indeed, since antiquity, it has been obtained from plant ashes brought into contact with water in which soluble compounds dissolve and in particular potassium carbonate, the resulting solution is then filtered and then placed in a pot, so that the water evaporates and potash is formed. This is a reference to the method of preparing potash. The name of the element 19, potassium, derives from the neo latin Potassa which itself derives from the German Potaschen formed from the association of the terms "Pot" and "aschen" meaning respectively "Pot" and "ashes". Potassium PropertiesĪlthough the spelling of potassium starts with P but the Potassium symbol is K, stange. Potassium is a chemical element that is numbered 19 in the Periodic Table of the Elements. 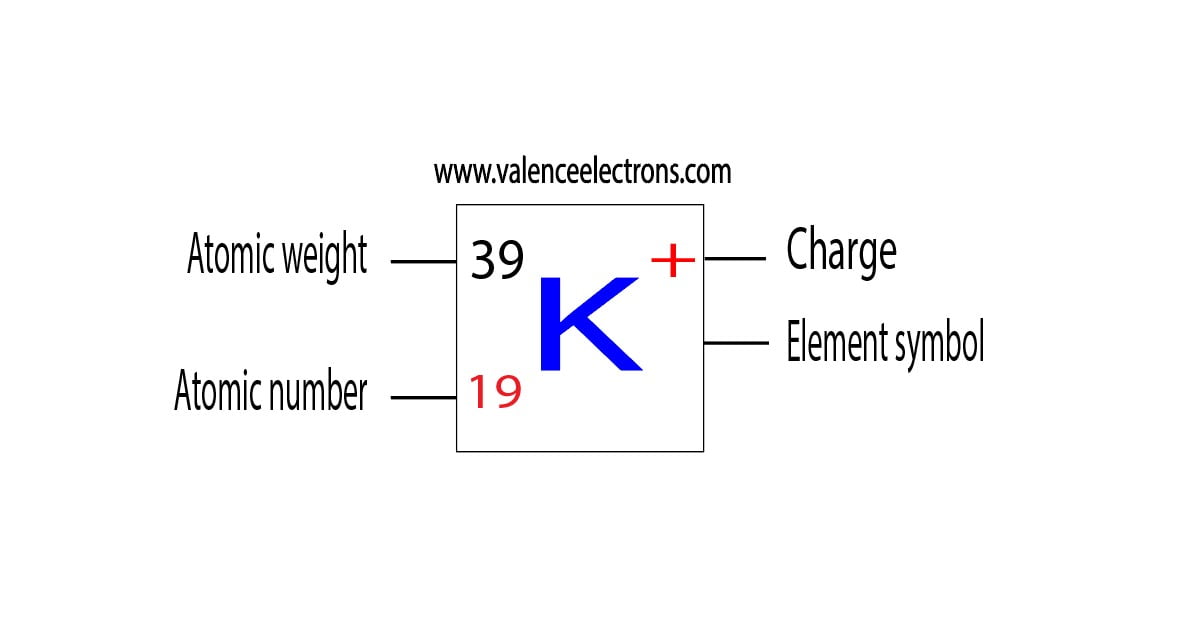
Sodium, have been known since antiquity and have been used in various fields of human activity. Potassium, like its closest chemical element i.e.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
